Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista de Ingeniería
Print version ISSN 0121-4993
rev.ing. no.27 Bogotá Jan./June 2008
Reproducible Fabrication of Scanning Tunneling Microscope Tips
Fabricación reproducible de puntas de barrido para microscopios de efecto túnel
Rodrigo Bernal
Ingeniero electrónico e Ingeniero mecánico. Facultad de Ingeniería, Universidad de los Andes. Bogotá D.C., Colombia.
go_rodrigo@yahoo.com
Alba Ávila
Ph.D. Profesora asistente, Departamento de Ingeniería Eléctrica y Electrónica. Investigadora en el Centro de Microelectrónica (CMUA), Facultad de Ingeniería, Universidad de los Andes. Bogotá D.C., Colombia.
Recibido 31 de agosto de 2007, aprobado 20 de febrero 2008.
PALABRAS CLAVES
Efecto túnel, microscopía, nanotecnología, procesos electroquímicos, tungsteno.
RESUMEN
La fabricación reproducible de puntas de barrido para microscopios de efecto túnel es demostrada usando un método electroquímico. Las puntas son hechas a partir de alambre de tungsteno de 0.5 mm de diámetro. Controlando las condiciones de fabricación: la inmersión en la solución electrolítica, rangos de voltaje-corriente y duración del proceso se logra realizar puntas con relaciones de aspecto de 0.68 y radios de punta de 330 nm en el mejor caso.
KEY WORDS
Electrochemical processes, microscopy, nanotechnology, tungsten, tunneling.
ABSTRACT
Reproducible fabrication of Scanning Tunneling Microscope tips is carried out following an electrochemical procedure widely reported. Tips are made from 0.5 diameter tungsten wire. Controlling fabrication parameters (immersion in the electrochemical solution, current-voltage ranges, duration of the process, among others) we achieve aspect ratios of 0.68 and tip radii of 330 nm in the best case.
INTRODUCTION
The scanning tunneling microscope (STM) is a widely- known and important surface science instrument, its capacity to perform atomic-scale characterization being the cause of this importance. The tunneling phenomenon in the microscope occurs when a conductive sample and a very sharp metallic tip are placed in near-contact separation (<1 nm) [1] and a voltage difference is applied between them.
From the very first experiments with the STM it is known that its resolution depends on the sharpness of the tips [1], [2], [3], with increasing sharpness directly leading to a better resolution (up to atomic resolution). This STM tips are fabricated mainly with Platinum-Iridium (Pt-Ir) or Tungsten(W) wires. Pt- Ir tips are made either by cutting or electrochemical methods, the former being more usual. On the other hand, tungsten tips are fabricated exclusively by electrochemical methods [4]. One of these techniques is the DC-drop off method [5].
In the Drop off method, a DC voltage is applied between two electrodes immersed in an electrolytic solution, the tungsten wire being the anode. Material is removed from the tungsten wire, once the voltage is applied between the anode and an extra electrode. If the electrochemical cell is properly constructed, the removal of material will cause the formation of a neck in the tungsten wire which will lead ultimately to its rupture, leaving a sharp tip in the zone where the fracture occurs.
The name of the method comes from the fact that when the wire breaks, the portion of the wire below the fracture zone drops into the electrochemical solution, while the upper part is used as the scanning tip.
Relevant parameters for this method have been shown to be the voltages in the reaction; which must make the process an electropolishing one [4]; the length of wire which drops (depth of immersion), which mainly defines aspect ratio of the tip (tip length over wire diameter); and the time between the drop-off and the removal of the voltage, called the cut-off time, which has an important effect on tip sharpness (tip-radius); shorter times lead to sharper tips [5].
In this work, the fabrication of STM tips is successfully carried out by this method. It is shown that reproducible results are possible with methodical control of the parameters above mentioned; the process is simple and could be easily adapted to other wire materials.
METHODOLOGY
The experimental set-up (electrochemical cell) used is schematically shown on Figure 1. Polycrystalline tungsten wire (Goodfellow 0.5 mm dia. 99.95% pure) is immersed to a depth li in KOH 2M. The depth of immersion is controlled and measured by the turning of a screw. In the surface of the liquid there is a copper wire (23AWG) bend in the form at a circular ring (8 mm dia.). Both ring and wire serve as electrodes in the electrochemical reaction, the wire being the anode.
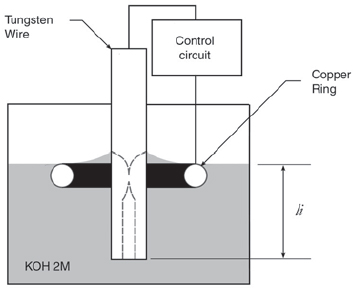
Figure 1. Schematic of the experimental setup for the electrochemical fabrication of STM tips. The dotted lines show schematically how the necking produces a tip in the upper portion of the wire.
The potential difference is applied by means of a control circuit (Figure 2) designed to monitorize the electropolishing current and also to provide appropriate cut-off times in order to obtain sharp tips. It is based on a design by Nakamura [6].
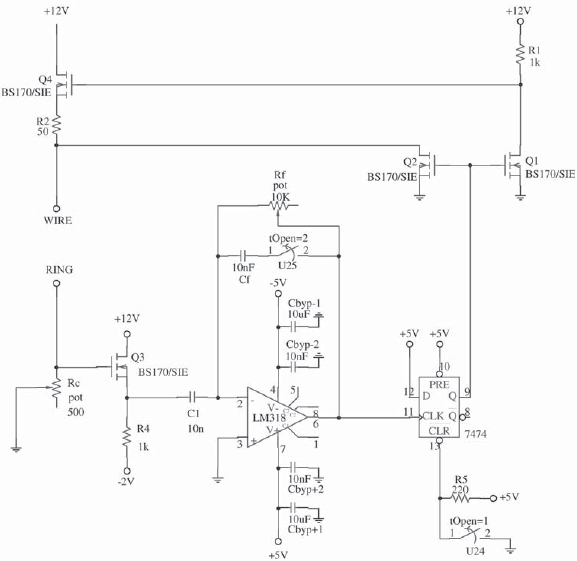
Figure 2. Control circuit used to monitorize the electropolishing current and to provide short cut-off times.
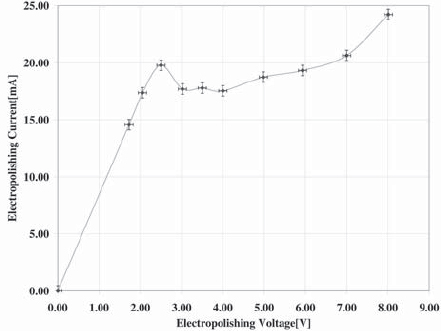
Figure 3. Voltagram of the electrochemical cell obtained with 2mm of depth of immersion.
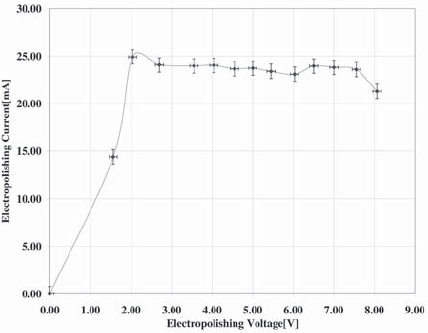
Figure 4. Voltagram of the electrochemical cell obtained with 3mm of depth of immersion.
In this circuit the electropolishing current is controlled by means of potentiometer Rc. The emitter-follower Q3 pass a voltage signal to a differentiator. This signal is analogous to the electropolishing current. When there is a sudden fall in this quantity, which indicates tip formation (rupture), the differentiator provides a peak of voltage which in turn changes the state of a flip-flop. The flip-flop turns on transistors Q1 and Q2, Q1 lowers the gate voltage of Q4, thus cutting the voltage provided to the electrochemical reaction. Cut-off times of 160 ns are typical.
To measure the effect of depth of immersion in tip morphology and radius, experiments for 2 mm and 3 mm are carried out. The respective voltagram (I-V curve) of each situation is obtained, and the reaction is performed in the optimum I-V ranges.
Tip radius and morphology is observed in a FEI Quanta 200 Scanning Electron Microscope (SEM).
RESULTS AND ANALYSIS
The voltagram of the electrochemical cell with 2 mm of tungsten wire immersed in the solution is shown on Figure 3, while the results for 3 mm are shown on Figure 4.
The electropolishing reaction is better carried out in the plateau region of the voltagram [7], leading to smoother surfaces and uniform removal of material, and in the lowest-possible voltage [5], thus lending lower oxide formation rates. Then, appropriate voltages for carrying out the electrochemical reactions are 2.5 V and 3 V for 2 mm and 3 mm depth of immersion respectively.
The voltagram for an immersion of 3 mm indicates that the currents are higher in this condition, which implies shorter fabrication times with increasing immersion depth. However, as is noted below, this is not always convenient.
Secondary electron SEM images of typical tips obtained by us are shown on Figures 5 and 6. Aspect ratio for 2 mm of immersion is consistently 0.68 (evaluated in five tips), with standard deviation 0.01. For 3 mm it is consistently 0.83 (evaluated again in five tips), with standard deviation 0.05. Then, 2 mm of immersion is a better condition, as the aspect ratio is lower, thus making the tip less susceptible to vibrations while operating in the STM. The increasing of aspect ratio by increasing depth of immersion has been observed also by other authors [5].
Both tips have good superficial quality, which shows that the voltage ranges used for the fabrication were appropriate. Also, they are highly symmetric (important for STM imaging [8]), showing the experimental set-up was correctly constructed. The reproducibility achieved is shown by the low standard deviation of the aspect ratio, obtained in both cases.

Figure 5. SEM image of tip morphology obtained with 2mm of immersion.

Figure 6. SEM image of tip morphology obtained with 3 mm of immersion.
A higher magnification of the tips can be used to measure tip radius. The micrographs are depicted on Figures 7 and 8. The results again show that 2 mm is a better option for depth of immersion as tip radius is 330 nm, compared to 456 nm obtained in the other case. Also, the sphere-like form of the 3 mm-tip indicates that the depth of immersion is excessive [5] as it causes a recoil of the material when the tip is formed.

Figure 7. SEM image of the tip-radius obtained with 2mm of immersion.

Figure 8. SEM image of the tip-radius obtained with 3mm of immersion.
A Back Scattered Electron SEM Image (Figure 9) of a 2 mm-tip shows a tungsten-oxide layer covering the surface of the tips [5]. This is a sub-product of the electrochemical reaction and thus occurs in every electropolishing experiment.

Figure 9. SEM image of a tip obtained with 2mm of immersion showing a non-conductive layer covering its surface.
Several methods have been reported in order to clean tips from this oxide. Under our available resources we find suitable to propose an Hydrofluoric Acid cleansing Procedure outlined by Hockett [9]. This method is yet to be implemented in our experiments. Effects of this cleaning procedure such as better stability of the tunneling junction have been reported [9].
CONCLUSIONS
Reproducible fabrication of STM tips was carried out, using procedures widely reported in literature. Satisfactory morphological and surface characteristics were obtained by using appropriate voltage ranges in the electrochemical process.
It was shown that 3 mm of depth of immersion is an excessive value, thus defining its upper limit. Better aspect ratio and tip radius were obtained by 2 mm depth of immersion.
However, tip-radius is still rather high as compared by that obtained by other authors [6]. Further research is underway to improve fabrication parameters, for example cut-off time to values as short as 50ns as reported by [6].
ACKNOWLEDGMENTS
We thank Universidad Nacional de Colombia (Bogotá D.C.) for the electron microscopy services.
REFERENCES
[1] J. Tersoff and D.R. Hamann. Theory of the scanning tunneling microscope. Physical Review B (Condensed Matter), Vol. 31, Jan. 1985, pp. 805–813. [ Links ]
[2] G. Binnig, H. Rohrer, C. Gerber and E. Weibel. Surface Studies by Scanning Tunneling Microscopy. Physical Review Letters, Vol. 49, Jul. 1982, pp. 57–61. [ Links ]
[3] P. K. Hansma and J. Tersoff. Scanning tunneling microscopy. Journal of Applied Physics, Vol. 61, Jan. 1987, pp. R1–R23. [ Links ]
[4] A. J. Melmed. The art and science and other aspects of making sharp tips. Journal of Vacuum Science & Technology B: Microelectronics and Nanometer Structures, Vol. 9, No. 2, Mar. 1991, pp. 601–608. [ Links ]
[5] J. P. Ibe, P. P. Bey, S. L. Brandow, R. A. Brizzolara, N. A. Burnham, D. P. Dilella, K. P. Lee, C. R. K. Marrian and R. J. Colton. On the electrochemical etching of tips for scanning tunneling microscopy. Journal of Vacuum Science Technology, Vol. 8, Jul. 1990, pp. 3570–3575. [ Links ]
[6] Y. Nakamura, Y. Mera, and K. Maeda. A reproducible method to fabricate atomically sharp tips for scanning tunneling microscopy. Review of Scientific Instruments, Vol. 70, Aug. 1999, pp. 3373–3376. [ Links ]
[7] G. V. Voort. Electrolytic Polishing. ASM Handbook, Ed. American Society for Metals, Vol. 9: Metallogrphy and Microstructures, 1993. [ Links ]
[8] A. I. Oliva, M. Aguilar, and V. Sosa. Low- and high-frequency vibration isolation for scanning probe microscopy. Measurement Science and Technology, Vol. 9, Mar. 1998, pp. 383–390. [ Links ]
[9] L. A. Hockett and S. Creager. A Convenient Method for Removing Surface Oxides from Tungsten STM Tips. Review of Scientific Instruments, Vol. 64, No. 1, 1993, pp. 263–264. [ Links ]